Full Field Optical Imaging
Optical coherence tomography (OCT)
Jean-Marie Chassot: engineer and head of the full-field OCT platform
To image scattering media in depth with a micrometric resolution, we develop optical tomography methods. Although full-field OCT practice is routine in the laboratory, this method has undergone recent developments, including:
Dynamic full-field OCT which provides an image of the metabolic activity of fresh tissue. This new contrast is a valuable tool for intra-operative diagnosis (Figure 1) or retinal cell activity (Figure 2).
Apelian C., Harms F., Thouvenin O. and A. C. Boccara, Biomed. Opt. Express 7, 1511-1524 (2016).
 Figure 1: Metabolic image of brain metastatic tissue where cancer cells are visible as well as their dynamics. Cells form lobules that are identified by the dense rounded shape of the structure.
Figure 1: Metabolic image of brain metastatic tissue where cancer cells are visible as well as their dynamics. Cells form lobules that are identified by the dense rounded shape of the structure.  Figure 2: Dynamic imaging of the ganglion cell layer of a monkey retina. (red=> blue fastest decorrelation). The ganglion cells and their nuclei are highlighted between the axons and the capillaries. The full-field OCT allows us to reveal without marking all the cells and cell types in the different layers of the retina.
Figure 2: Dynamic imaging of the ganglion cell layer of a monkey retina. (red=> blue fastest decorrelation). The ganglion cells and their nuclei are highlighted between the axons and the capillaries. The full-field OCT allows us to reveal without marking all the cells and cell types in the different layers of the retina.
An aberration-insensitive resolution: We have shown experimentally and theoretically that the use of spatially incoherent light led to an impulse response whose width was independent of the level of aberrations. This spectacular and unexpected result paves the way for high-resolution retinal imaging that can do without adaptive optics.
P Xiao, M. Fink, and A. C. Boccara, Journal of Biomedical Optics 21(12), 121505 (2016)
A noise-induced mechanical contrast: In line with what has been done in helio-seismology or acoustics, we have shown how to go back to the local mechanical properties of tissues excited by a random and isotropic mechanical noise (such as internal movements of the human body).
T-M Nguyen, A. Zorgani ; M. Lescanne ; C. Boccara ;M. Fink ; .S. Catheline, Diffuse shear wave imaging: toward passive elastography using low-frame rate spectral-domain optical coherence tomography J. Biomed. Opt. 21(12), 126013 (Dec 21, 2016).
Deep tissue access in vivo - Biometry and Ophthalmology: Original experimental devices associated with the use of optimized cameras have allowed access to in vivo studies. In the case of highly scattering tissues, such as the skin, the image of so-called "internal" fingerprints located a few hundred microns below the surface was obtained and confirmed by the use of our setup by 2 foreign laboratories (Figure 3) . In contrast, a medium with very low diffusion, such as the cornea, could be observed without contact with cellular resolution (Figure 4).
E. Auksorius, and A. C. Boccara, "Fingerprint imaging from the inside of a finger with full-field optical coherence tomography," Biomed. Opt. Express 6(11), 4465-4471 (2015).
V. Mazlin, E. Dalimier, K.F. Grieve, et al. “Non-contact full-field optical coherence tomography: a novel tool for in vivo imaging of the human cornea (Conference Presentation)”, Proc. SPIE 10045, Ophthalmic Technologies XXVII, 1004519 (2017)
 Figure 3: Fingerprints after surface of a fingertip was damaged by rubbing it with sandpaper. The quality of the surface (conventional) fingerprint image (left) is significantly reduced, whereas the internal fingerprint image (right) is highly contrasted.
Figure 3: Fingerprints after surface of a fingertip was damaged by rubbing it with sandpaper. The quality of the surface (conventional) fingerprint image (left) is significantly reduced, whereas the internal fingerprint image (right) is highly contrasted.
 Figure 4: Full-field OCT image of the human cornea in vivo. Layer depth is marked in red. Corneal layers, located at different depths (from 0 µm to 140 µm deep), are seen in a single en face image due to the curvy shape of the cornea.
Figure 4: Full-field OCT image of the human cornea in vivo. Layer depth is marked in red. Corneal layers, located at different depths (from 0 µm to 140 µm deep), are seen in a single en face image due to the curvy shape of the cornea.
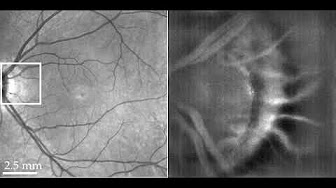
 Figure 5: In vivo human retinal imaging of near periphery at 6° eccentricity inferior to the foveal center with a field of view of 2.4°×2.4°. (a) The Spectral Domain OCT (SDOCT) cross-sectional image of the imaging position with the red (RNFL: retinal nerve fiber layer) and blue (IS/OS: inner/outer segment of photoreceptors layer) dashed lines indicating the FFOCT imaging depth. (b) Fundus photography with the black box indicating the FFOCT imaging area and the green dashed line showing the SDOCT scanning position. (c-d) In vivo Full Field OCT image of at 6° inferior to the fovea, at the RNFL (c) and IS/OS photoreceptor layers (d) without Adaptive Optics (AO). (e) The 2D power spectra of (d) showing the Yellot’s ring, the radius of which is related to the cone photoreceptor spacing. (f-h) The AO retinal camera image around the RNFL (f) and IS/OS photoreceptor layer (g) at the same retinal location and the 2D power spectra (h) of (g). Scale bar: 100 µm..
Figure 5: In vivo human retinal imaging of near periphery at 6° eccentricity inferior to the foveal center with a field of view of 2.4°×2.4°. (a) The Spectral Domain OCT (SDOCT) cross-sectional image of the imaging position with the red (RNFL: retinal nerve fiber layer) and blue (IS/OS: inner/outer segment of photoreceptors layer) dashed lines indicating the FFOCT imaging depth. (b) Fundus photography with the black box indicating the FFOCT imaging area and the green dashed line showing the SDOCT scanning position. (c-d) In vivo Full Field OCT image of at 6° inferior to the fovea, at the RNFL (c) and IS/OS photoreceptor layers (d) without Adaptive Optics (AO). (e) The 2D power spectra of (d) showing the Yellot’s ring, the radius of which is related to the cone photoreceptor spacing. (f-h) The AO retinal camera image around the RNFL (f) and IS/OS photoreceptor layer (g) at the same retinal location and the 2D power spectra (h) of (g). Scale bar: 100 µm..
Finally for detection of nanoparticles (eg. viruses and vesicles) we have developed and patented a sensitive method (based on a sensitive "common path" interferometric detection of scattered light allowing to follow their Brownian motion in a liquid environment A startup is being created on this subject.
M. Boccara, Y. Fedala, C. Venien Bryan, M. Bailly-Bechet, C. Bowler, and A.C. Boccara, “Full-field interferometry for counting and differentiating aquatic biotic nanoparticles: from laboratory to Tara Oceans”, Biomedical Optics Express Vol. 7,Issue 9, pp. 3736-3746 (2016).
Patent : Méthode et dispositif de détection optique de nanoparticules dans un échantillon fluide, WO 2016055306 A1 2015 Albert Claude Boccara, Martine Boccara. (Brevet Français + PCT)
Digital holography
Our holographic imaging instruments are designed and optimized for high-speed measurements in very low light. Their applications range from vibrometry to micro-rheology. The visualization of images acquired by holographic interferometry requires a calculation of optical wave propagation. We have developed Holovibes, a holographic image rendering software in real-time by graphics card (GPU) calculations. This software has been validated for Doppler laser imaging, vibrometry, microscopy, and OCT.
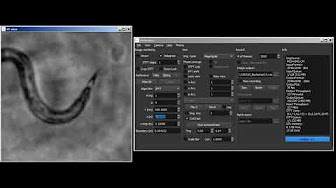 Real-time rendering of Gabor holograms of C. elegans worms in water by propagation of the angular spectrum of interferograms recorded with a common-path digital holographic microscope.
Real-time rendering of Gabor holograms of C. elegans worms in water by propagation of the angular spectrum of interferograms recorded with a common-path digital holographic microscope.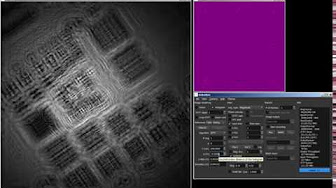 Real-time rendering of digital Gabor holograms of an absorptive USAF 1951 resolution target by propagation of the angular spectrum of interferograms recorded with a digital holographic microscope (DHM).
Real-time rendering of digital Gabor holograms of an absorptive USAF 1951 resolution target by propagation of the angular spectrum of interferograms recorded with a digital holographic microscope (DHM).
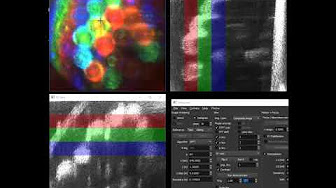 Real-time holographic swept-source OCT of a sample of 1.5 mm silica beads wrapped with tape. A composite XY image is displayed by additive color from RGB frequency bands.
Real-time holographic swept-source OCT of a sample of 1.5 mm silica beads wrapped with tape. A composite XY image is displayed by additive color from RGB frequency bands. Real-time hologram rendering and short-time Fourier transformation (STFT) for imaging transient vibrations in a guitar.
Real-time hologram rendering and short-time Fourier transformation (STFT) for imaging transient vibrations in a guitar.
Surface acoustic waves imaging We have developed a method for narrow-band imaging of surface acoustic waves by tunable stroboscopic holography. The originality is to use a multiplexed local oscillator to process several optical sidebands in the time bandwidth of a camera. This method allows the absolute measurement of vibration amplitudes with a sub-nanometric sensitivity and can be used, for example, to demonstrate local delamination in composite sandwich plates.
Holographic imaging of surface acoustic waves, F. Bruno, J. Laurent, D. Royer, M. Atlan, Applied Physics Letters 104, no. 8 (2014).
Holographic Doppler Laser Imaging of Blood Flow With a video camera, holography allows selective frequency imaging in the low frequency range in combination with stroboscopic lighting. Microvascular hemodynamic imaging has been demonstrated in rodents with laser light without an exogenous marker.
Holographic laser Doppler imaging of microvascular blood flow, C. Magnain, A. Castel, T. Boucneau, M. Simonutti, I. Ferezou, A. Rancillac, T. Vitalis, J.A. Sahel, M. Paques, M. Atlan, JOSA A, Vol. 31, Issue 12, pp. 2723-2735 (2014).
 Lateral motion compensation from intensity-based registration of digital holograms of the retina reconstructed in real time by Fresnel transform.
Lateral motion compensation from intensity-based registration of digital holograms of the retina reconstructed in real time by Fresnel transform.
Ultrahigh-speed imaging of microvascular blood flow has also been demonstrated, up to an image acquisition rate of 75 kHz. We have found that contrasts of local dynamics emerge from envelopes of fast Fourier transforms of holograms and their inter-correlations. This approach allows imaging of blood flow in the retinal and brain vessels.
High speed optical holography of retinal blood flow, M. Pellizzari, M. Simonutti, J. Degardin, J.-A. Sahel, M. Fink, M. Paques, M. Atlan, Optics Letters Vol. 41, Issue 15, pp. 3503-3506 (2016).
In vivo laser Doppler holography of the human retina. Puyo, L., et al. Biomedical optics express 9.9 (2018): 4113-4129